Change in molar entropy during a first-order phase transition¶
Code: #11D-000
File: apps/van_der_waals/entropy.ipynb
The aim of this notebook is to visualize the change in molar entropy during a first-orden liquid-gas transition.
Interface¶
The main interface (main_block_11D_000) is divided in two HBox: top_block_11D_000 and bottom_block_11D_000. bottom_block_11D_000 contains of 2 bqplot Figures: fig_11D_001 and fig_11D_002.
[1]:
from IPython.display import Image
Image(filename='../../static/images/apps/11D-000_1.png')
[1]:
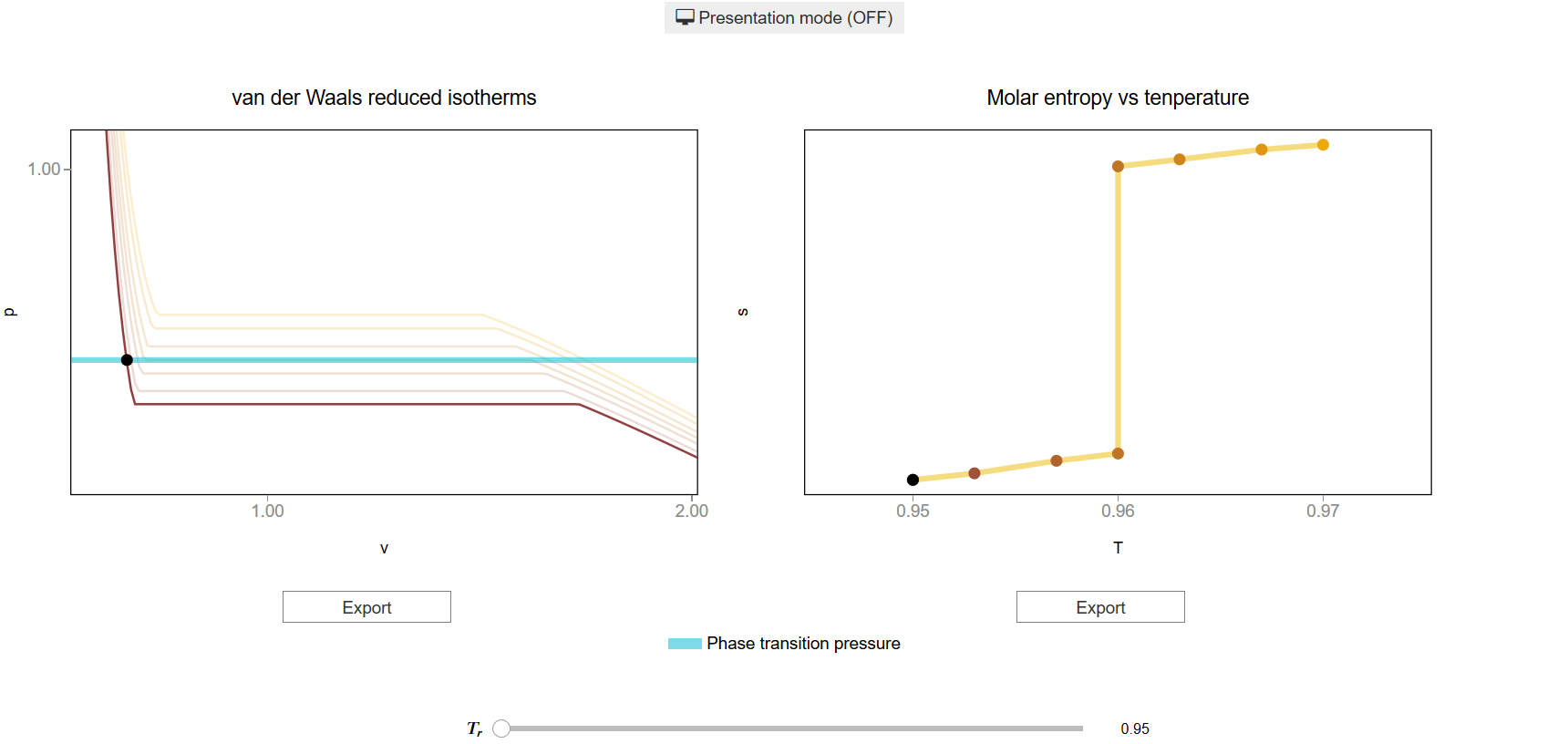
The slider T_slider updates the values of \(T\) which updates the lines and scatter points of fig_11D_001 and fig_11D_002.
[2]:
Image(filename='../../static/images/11D-000_2.png')
[2]:

CSS¶
A custom css file is used to improve the interface of this application. It can be found here.
[3]:
from IPython.display import HTML
display(HTML("<head><link rel='stylesheet' type='text/css' href='./../../static/custom.css'></head>"))
display(HTML("<style>.container { width:100% !important; } .jupyter-button {white-space: normal !important;}</style>"))
Packages¶
[4]:
from bqplot import *
import bqplot as bq
import bqplot.marks as bqm
import bqplot.scales as bqs
import bqplot.axes as bqa
import ipywidgets as widgets
from scipy.signal import argrelextrema
import urllib.parse
import webbrowser
import sys
Physical functions¶
This are the functions that have a physical meaning:
get_relative_isothermsexperimetal_isothermsget_rootsp_indefinite_integralp_definite_integralfind_real_fixed_pfind_real_fixed_T
[5]:
def get_relative_isotherms(v_range, T_range):
"""This function calculates the theoretical p(v, T) plane
(in reduced coordinates) according to van der Waals
equation of state from a given range of volumes
and tenperatures.
Args:
v_range: An array containing the values of v
(in reduced coordinates)for which the isotherms must be
calculated.\n
T_range: An array containing the values of T
(in reduced coordinates)for which the isotherms must be
calculated.\n
Returns:
isotherms: A list consisted of numpy arrays containing the
pressures of each isotherm.
"""
isotherms = []
for T in T_range:
p_R = []
for v in v_range:
val = (8.0/3.0*T/(v - 1.0/3.0) - 3.0/v**2)
p_R = np.append(p_R, val)
isotherms.append(p_R)
return isotherms
[6]:
def experimental_isotherms(p_range, v_range, T_range, fixed_p, fixed_T):
"""This function calculates the experimental p(v, T) plane
(in reduced coordinates) according to van der Waals
equation of state for a given range of volumes
and tenperatures or for a given range of volumes
and pressures.
Args:
p_range: An array containing the values of p
(in reduced coordinates)for which the isotherms must be
calculated. Only used if fixed_p == True.\n
v_range: An array containing the values of v
(in reduced coordinates)for which the isotherms must be
calculated.\n
T_range: An array containing the values of v
(in reduced coordinates)for which the isotherms must be
calculated. Only used if fixed_T == True.\n
fixed_p: Boolean variable which represents if the isotherms
must be calculated for a given pressures.\n
fixed_T: Boolean variable which represents if the isotherms
must be calculated for a given pressures.\n
Returns:
expe_data: A list consisted of numpy arrays containing the
pressures of each theoretical isotherm.\n
theo_data: A list consisted of numpy arrays containing the
pressures of each theoretical isotherm.\n
v_limits: A list consisted of arrays of the volume limits of
the phase-transition of each subcritical isotherm.\n
p_limits: A list consisted of arrays of the pressure limits of
the phase-transition of each subcritical isotherm.\n
tenperatures: A list consisted of the tenperatures of the
isotherms.\n
"""
if fixed_T:
theo_data = get_relative_isotherms(v_range, T_range)
expe_data = []
v_limits = []
p_limits = []
p_range = np.linspace(0.001, 1.0, num=10000)
pressures, v_isobaric_limits = find_real_fixed_T(p_range, T_range)
for i in range(len(theo_data)):
p_expe = []
if i < len(v_isobaric_limits):
v_lim = v_isobaric_limits[i]
if len(v_lim) > 1: #check if there is only one point
for j in range(len(v_range)):
if v_range[j] > v_lim[0] and v_range[j] < v_lim[1]:
p_expe.append(pressures[i])
else:
p_expe.append(theo_data[i][j])
v_limits = np.append(v_limits, [v_lim[0], v_lim[1]])
p_limits = np.append(p_limits, [pressures[i], pressures[i]])
else:
p_expe = theo_data[i]
v_limits = np.append(v_limits, [1.0])
p_limits = np.append(p_limits, [1.0])
else:
p_expe = theo_data[i]
expe_data.append(p_expe)
tenperatures = T_range
return expe_data, theo_data, p_limits, v_limits, tenperatures
elif fixed_p:
tenperatures, v_isobaric_limits = find_real_fixed_p(p_range, T_range)
theo_data = get_relative_isotherms(v_range, tenperatures)
expe_data = []
v_limits = []
p_limits = []
for i in range(len(theo_data)):
p_expe = []
if i < len(v_isobaric_limits):
v_lim = v_isobaric_limits[i]
if len(v_lim) > 1: #check if there is only one point
for j in range(len(v_range)):
if v_range[j] > v_lim[0] and v_range[j] < v_lim[1]:
p_expe.append(p_range[i])
else:
p_expe.append(theo_data[i][j])
v_limits = np.append(
v_limits,
[v_lim[0],
v_lim[1]]
)
p_limits = np.append(
p_limits,
[p_range[i],
p_range[i]]
)
else:
p_expe = theo_data[i]
v_limits = np.append(v_limits, [1.0])
p_limits = np.append(p_limits, [1.0])
else:
p_expe = theo_data[i]
expe_data.append(p_expe)
return expe_data, theo_data, p_limits, v_limits, tenperatures
[7]:
def get_roots(p, T):
"""This function calculates the roots of a van der Waals
isotherm of a given T and set of pressures.
Args:
p: Numpy array consisted of the pressures of the isotherm.\n
T: Value of the tenperature.\n
Returns:
roots_in_range: A list consisted of the real roots.\n
"""
roots = np.roots([1.0, - 1.0/3.0*(1.0 + 8.0*T/p), 3.0/p, -1.0/p])
roots_in_range = []
for root in roots:
if np.isreal(root):
root = np.real(root)
if root > 0:
roots_in_range.append(root)
roots_in_range.sort()
return roots_in_range
[8]:
def p_indefinite_integral(p_0, v_0, T):
"""This function calculates the indefinite integral between
a van der Waals isotherm and a isobaric line.
Args:
p0: Isobaric line's pressure.\n
v0: Value of the volume.\n
T: Value of the tenperature.\n
Returns:
integral: Value of the indefinite integral between a
van der Waals isotherm at T and a isobaric line of p0 at a
volume v0.\n
"""
integral = 8.0/3.0 * T *np.log(v_0 - 1.0/3.0) + 3.0/v_0 - p_0*v_0
return integral
[9]:
def definite_integral(p_0, v_range, T):
"""This function 'p_indefinite_integral' function to calculate
the definite integral between a van der Waals isotherm and a
isobaric line.
Args:
p0: Isobaric line's pressure.\n
v_range: Tuple or list consisted of volume limits.\n
T: Value of the tenperature.\n
Returns:
integral: Value of the definite integral between a
van der Waals isotherm at T and a isobaric line of p0 in a
volume range v_range.\n
"""
v_0, v_1 = v_range[0], v_range[1]
integral = p_indefinite_integral(p_0, v_1, T) - p_indefinite_integral(p_0, v_0, T)
return integral
[10]:
def find_real_fixed_T(p_values, T_values):
"""This function uses Maxwell's construction to find the
pressures in which phase transition happens given some
fixed tenperatures.\n
Args:
p_values: List of pressures in which the real isotherm is
searched.\n
T_values: List of tenperatures of the isotherms.\n
Returns:
pressures: List of pressures in which phase transition
happens.\n
v_range: Volume limits of phase transition zones.
"""
eps = 1e-3
pressures = []
v_ranges = []
for T in T_values:
if T < 1.0:
for p in p_values:
roots = get_roots(p, T)
if len(roots) == 3:
v_range = [roots[0], roots[2]]
area = definite_integral(p, v_range, T)
if abs(area) < eps:
pressures.append(p)
v_ranges.append(v_range)
break
elif T == 1.0:
pressures.append(1.0)
v_ranges.append([1.0])
return pressures, v_ranges
[11]:
def find_real_fixed_p(p_values, T_values):
"""This function uses Maxwell's construction to find the
tenperatures in which phase transition happens given some
fixed pressures.\n
Args:
p_values: List of pressures of the isotherms.\n
T_values: List of tenperatures in which the real isotherm is
searched.\n
Returns:
tenperatures: List of tenperatures in which phase transition
happens.\n
v_range: Volume limits of phase transition zones.
"""
eps = 1e-3
tenperatures = []
v_ranges = []
for p in p_values:
if p < 1.0:
for T in T_values:
roots = get_roots(p, T)
if len(roots) == 3:
v_range = [roots[0], roots[2]]
area = definite_integral(p, v_range, T)
if abs(area) < eps:
tenperatures.append(T)
v_ranges.append(v_range)
break
elif p == 1.0:
tenperatures.append(1.0)
v_ranges.append([1.0])
return tenperatures, v_ranges
[12]:
def get_entropy(v_values):
"""This function calculates the entropy for a given
array of molar volumes.
Args:
v_values: Array consisted of the values of molar volume.
Returns:
s: Array containing the values of the entropy.
"""
v_values = np.asarray(v_values)
s = 8.0/3.0*np.log(3.0*v_values - 1.0)
return s
Main interface¶
[ ]:
T_values = np.round(np.linspace(0.95, 0.97, 7), 3)
v_values = np.linspace(0.45, 5.2, 500)
colors = generate_gradient('#914040', '#feb901', len(T_values))
def_op = 0.2
opacities = [def_op for T in T_values]
opacities[0] = 1.0
data = experimental_isotherms(
p_range=[],
v_range=v_values,
T_range=T_values,
fixed_T = True,
fixed_p = False
)
expe_p_values = data[0]
theo_p_values = data[1]
p_limits = data[2]
v_limits = data[3]
T_limits = data[4]
# The index of the pressure of the selected isotherm
selected = 3
p_0 = np.unique(p_limits)[selected]
state_v = []
T_values_repeated = []
colors_repeated = []
for i in range(len(T_values)):
if i == selected:
state_v.append(
np.array([v_limits[2*i],v_limits[2*i+1]])
)
T_values_repeated.append(
T_values[i]
)
colors_repeated.append(colors[i])
else:
idx = find_nearest_index(expe_p_values[i], p_0)
state_v.append(v_values[idx])
T_values_repeated.append(
T_values[i]
)
colors_repeated.append(colors[i])
s_values = []
for v in state_v:
s_values.append(get_entropy(v))
s_values_plain = np.hstack(s_values)
##########################
########TOP BLOCK#########
##########################
top_block_11D_000 = widgets.HBox(
[],
layout=widgets.Layout(
align_items='center',
width='100%'
)
)
# fig_11D_001
scale_x_11D_001 = bqs.LinearScale(min = 0.55, max = 2.0)
scale_y_11D_001 = bqs.LinearScale(min = 0.75, max = 1.02)
axis_x_11D_001 = bqa.Axis(
scale=scale_x_11D_001,
tick_format='0.2f',
tick_style={'font-size': '15px'},
tick_values=[1.0, 2.0, 3.0, 4.0, 5.0],
grid_lines = 'none',
grid_color = '#8e8e8e',
label='v',
label_location='middle',
label_style={'stroke': 'black', 'default-size': 35},
label_offset='50px'
)
axis_y_11D_001 = bqa.Axis(
scale=scale_y_11D_001,
tick_format='0.2f',
tick_style={'font-size': '15px'},
tick_values=[0.0, 1.0],
grid_lines = 'none',
grid_color = '#8e8e8e',
orientation='vertical',
label='p',
label_location='middle',
label_style={'stroke': 'red', 'default_size': 35},
label_offset='50px'
)
isotherms = bqm.Lines(
x = np.array([v_values]),
y = np.array([expe_p_values]),
scales = {'x': scale_x_11D_001, 'y': scale_y_11D_001},
opacities = opacities,
visible = True,
colors = colors,
labels = [str(t) for t in T_limits],
)
isobaric = bqm.Lines(
x = [v_values[0], v_values[-1]],
y = np.array([p_0, p_0]),
scales = {'x': scale_x_11D_001, 'y': scale_y_11D_001},
opacities = [0.5],
visible = True,
colors = ['#01b6cf'],
stroke_width = 5,
)
state = bqm.Scatter(
x = [state_v[0]],
y = [p_0],
scales = {'x': scale_x_11D_001, 'y': scale_y_11D_001},
visible = True,
colors = ['black'],
names = [],
#tooltip = tt
)
fig_11D_001 = bq.Figure(
title='van der Waals reduced isotherms',
marks=[],
axes=[axis_x_11D_001, axis_y_11D_001],
animation_duration=0,
layout = widgets.Layout(
align_self='center',
),
legend_location='top-right',
background_style= {'fill': 'white','stroke': 'black'},
fig_margin=dict(top=80,bottom=80,left=60,right=30),
toolbar = True
)
fig_11D_001.marks = [
isotherms,
isobaric,
state
]
# fig_11D_002
scale_x_11D_002 = bqs.LinearScale(min=0.945, max=0.975)
scale_y_11D_002 = bqs.LinearScale()
axis_x_11D_002 = bqa.Axis(
scale=scale_x_11D_002,
tick_format='0.2f',
tick_style={'font-size': '15px'},
tick_values=[0.95, 0.96, 0.97],
grid_lines = 'none',
grid_color = '#8e8e8e',
label='T',
label_location='middle',
label_style={'stroke': 'black', 'default-size': 35},
label_offset='50px'
)
axis_y_11D_002 = bqa.Axis(
scale=scale_y_11D_002,
tick_format='0.2f',
tick_style={'font-size': '15px'},
num_ticks=0,
grid_lines = 'none',
grid_color = '#8e8e8e',
orientation='vertical',
label='s',
label_location='middle',
label_style={'stroke': 'red', 'default_size': 35},
label_offset='50px'
)
entropy_line = bqm.Lines(
x = T_values_repeated,
y = s_values_plain,
scales = {'x': scale_x_11D_002, 'y': scale_y_11D_002},
opacities = [0.5],
visible = True,
colors = ['#edba00'],
labels = [str(t) for t in T_limits],
stroke_width = 5,
)
entropy_scatter = bqm.Scatter(
x = T_values_repeated,
y = s_values_plain,
scales = {'x': scale_x_11D_002, 'y': scale_y_11D_002},
opacities = opacities,
visible = True,
colors = colors_repeated,
labels = [str(t) for t in T_limits],
)
state_s = bqm.Scatter(
x = [T_values[0]],
y = [s_values_plain[0]],
scales = {'x': scale_x_11D_002, 'y': scale_y_11D_002},
visible = True,
colors = ['black'],
names = [],
)
fig_11D_002 = bq.Figure(
title='Molar entropy vs tenperature',
marks=[],
axes=[axis_x_11D_002, axis_y_11D_002],
animation_duration=0,
layout = widgets.Layout(
align_self='center',
),
legend_location='top-right',
background_style= {'fill': 'white','stroke': 'black'},
fig_margin=dict(top=80,bottom=80,left=60,right=30),
toolbar = True
)
fig_11D_002.marks = [
entropy_line,
entropy_scatter,
state_s
]
prepare_export_fig_11D_001_button = widgets.Button(
description='Export',
disabled=False,
button_style='',
tooltip='',
layout = widgets.Layout(
align_self='center'
)
)
prepare_export_fig_11D_001_button.on_click(prepare_export)
prepare_export_fig_11D_002_button = widgets.Button(
description='Export',
disabled=False,
button_style='',
tooltip='',
layout = widgets.Layout(
align_self='center'
)
)
prepare_export_fig_11D_002_button.on_click(prepare_export)
top_block_11D_000.children = [
widgets.VBox([
fig_11D_001,
prepare_export_fig_11D_001_button
]),
widgets.VBox([
fig_11D_002,
prepare_export_fig_11D_002_button
])
]
bottom_block_11D_000 = widgets.VBox(
[],
layout=widgets.Layout(
align_items='center',
width='100%'
)
)
isobaric_line = widgets.HTML(
value="<div style='width:30px;text-align:left;display:inline-block;" \
+ "border: 5px solid #01b6cf;opacity: 0.5'> </div>" \
+ " Phase transition pressure"
)
T_slider = widgets.SelectionSlider(
options=T_values,
value=T_values[0],
description=r'\( T_r \)',
disabled=False,
continuous_update=True,
orientation='horizontal',
readout=True,
layout = widgets.Layout(
width = '50%',
margin = '45px 0 0 0'
)
)
T_slider.observe(change_tenperature, 'value')
bottom_block_11D_000.children = [
isobaric_line,
T_slider
]
change_view_button = widgets.ToggleButton(
value=False,
description='Presentation mode (OFF)',
disabled=False,
button_style='',
tooltip='',
icon='desktop',
layout=widgets.Layout(
width='initial',
align_self='center'
)
)
change_view_button.observe(change_view, 'value')
main_block_11D_000 = widgets.VBox(
[],
layout=widgets.Layout(
align_items='center',
width='100%'
)
)
main_block_11D_000.children = [
change_view_button,
top_block_11D_000,
bottom_block_11D_000,
]
figures = [
fig_11D_001,
fig_11D_002
]
main_block_11D_000